NOTICE
ANSM - Updated on : 27/05/2025
Product name
MYCOSTER 1 per cent, cream
Ciclopirox olamine
Box
Please read this leaflet carefully before using this medicine, as it contains important information for you.
Always use this medicine exactly as it is described in this leaflet or by your doctor or pharmacist.
- Keep this leaflet. You may need to read it again.
- Ask your pharmacist for any advice or information you may need.
- If you experience any side effects, talk to your doctor or pharmacist. This also applies to any side effects not mentioned in this leaflet. See section 4.
- You should contact your doctor if you do not feel any improvement or if you feel less well.
What does this leaflet contain?
1. What is MYCOSTER 1 per cent cream and what is it used for?
2. What do I need to know before using MYCOSTER 1 percent cream?
3. How do I use MYCOSTER 1 per cent cream?
4. What are the possible side effects?
5. How should MYCOSTER 1 per cent cream be stored?
6. Package contents and other information.
1. WHAT IS MYCOSTER 1 per cent, cream AND WHAT IS IT USED FOR?
Pharmacotherapeutic class: topical antifungal agents - ATC code: D01AE14.
This medicine contains the active substance ciclopirox olamine, a cutaneous antifungal agent.
It is indicated for the treatment of
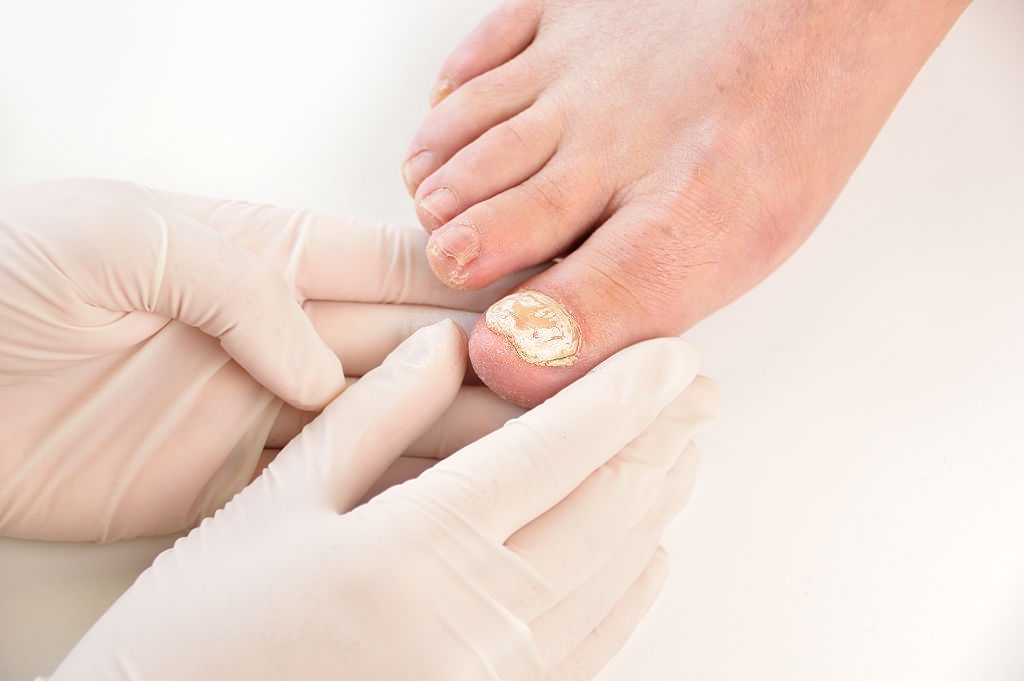
- fungal infections (mycoses) of the skin (dermatoses with or without bacterial superinfection) or nails (onychomycoses),
- mild to moderate seborrheic dermatitis of the face.
2. WHAT DO I HAVE TO KNOW BEFORE USING MYCOSTER 1 per cent, cream?
Never use MYCOSTER 1 per cent, cream:
- if you are allergic to ciclopirox olamine or any of the other ingredients listed in section 6.
Warnings and precautions
Talk to your doctor or pharmacist before using MYCOSTER.
- Do not swallow.
- Do not apply to open wounds.
- Avoid contact with eyes and mucous membranes.
- Candidiasis (fungal infections): do not use soaps with an acidic pH, as they encourage the multiplication of candida (fungus).
- In the event of an allergic reaction, stop treatment immediately, carefully remove MYCOSTER and contact a doctor.
Children
Not applicable.
Other medicinal products and MYCOSTER 1 per cent, cream
Tell your doctor or pharmacist if you are taking, have recently taken or may take any other medicines.
MYCOSTER 1 percent cream with food and drink
Not applicable.
Pregnancy and breast-feeding
If you are pregnant or breast-feeding, think you may be pregnant or are planning to become pregnant, ask your doctor or pharmacist for advice before taking this medicine.
Pregnancy
There are insufficient data on the use of ciclopirox olamine in pregnant women.
As a precautionary measure , MYCOSTERshould not be used during pregnancy.
Lactation
There are no adequate data on the excretion of ciclopirox olamine into breast milk.
As a precautionary measure, MYCOSTER should not be used during breast-feeding.
Driving and use of machinery
MYCOSTER has no effect on the ability to drive vehicles and use machines.
MYCOSTER 1 per cent, cream contains
This medicine contains :
- cetyl alcohol and stearyl alcohol, which may cause local skin reactions (e.g. contact dermatitis).
- 10 mg benzyl alcohol (E1519) per gram of cream, i.e. 300 mg per tube. Benzyl alcohol may cause allergic reactions and, on topical application, mild local irritation.
- contains polysorbate 60 (E435), which may cause allergic reactions.
3. HOW TO USE MYCOSTER 1 per cent, cream?
Always use this medicine exactly as instructed in this leaflet or as directed by your doctor or pharmacist. Check with your doctor or pharmacist if in doubt.
Dosage
Skin mycoses: 2 applications daily for approximately 21 days.
Nail fungus: apply cream alone or in combination with another treatment for several months.
Mild to moderate seborrheic dermatitis of the face:
- Attack treatment (2 to 4 weeks): 2 applications per day,
- Beyond that, as a maintenance treatment: 1 application per day for 28 days.
Method and route of administration
Cutaneous route.
For external use only.
Clean and dry areas to be treated.
Wash hands thoroughly before and after each application.
Apply MYCOSTER to the areas of skin to be treated and massage gently.
If you have used more MYCOSTER 1 per cent, cream than you should have, contact your doctor.
Not applicable.
If you forget to use MYCOSTER 1 per cent, cream
Do not use a double dose to make up for the one you forgot to apply.
If you stop using MYCOSTER 1 percent cream
Not applicable.
If you have any further questions about the use of this medicine, please ask your doctor or pharmacist.
4. WHAT ARE THE POSSIBLE SIDE EFFECTS?
Like all medicines, this drug may cause undesirable effects, but they do not occur systematically in everyone.
Common side effects (may affect up to one in 10 people):
- Temporary worsening (for a limited time) of certain local adverse reactions at the application site such as redness (erythema), itching (pruritus), which is generally transient and does not require discontinuation of treatment.
- Burning sensation.
These side effects may appear after the first applications. If these symptoms persist, consult a physician.
Uncommon side effects (may affect up to one person in 100)
- Local blister-like reactions.
- Generalized allergic reactions (hypersensitivity), which may be severe, in which you may experience swelling (edema), itching (pruritus), an itchy or non-itchy rash (urticaria) or difficulty breathing (dyspnea). If these effects occur, you should stop taking this drug immediately and contact a doctor as soon as possible.
Undetermined frequency (cannot be estimated on the basis of available data)
- Contact dermatitis (eczema-like rash).
If you apply MYCOSTER over a long period of time to a large surface area, to injured skin, to mucous membranes or under an occlusive (covering) dressing, the effects associated with the passage of the drug into the bloodstream should be taken into consideration : seek medical advice.
Reporting side effects
If you experience any side effects, please tell your doctor or pharmacist. This also applies to any side effects not mentioned in this leaflet. You can also report adverse reactions directly via the national reporting system: Agence nationale de sécurité du médicament et des produits de santé (ANSM) and the network of Centres Régionaux de Pharmacovigilance - Website: https://signalement.social-sante.gouv.fr/.
By reporting adverse reactions, you are helping to provide more information on drug safety.
5. HOW TO CONSERVE MYCOSTER 1 per cent, cream?
Keep out of sight and reach of children.
Do not use this medicine after the expiration date indicated on the tube after EXP. The expiry date refers to the last day of the month.
Before opening: Store at a temperature not exceeding 30°C.
After first opening the tube, this medicine should be stored for a maximum of 3 months.
Medicines must not be disposed of in the sewage system or with household waste. Ask your pharmacist what to do with unused medicines. These measures will help protect the environment.
6. CONTENTS AND OTHER INFORMATION
What MYCOSTER 1 per cent cream contains
- The active substance is :
Ciclopirox olamine......................................................................................................................1,000 g
Per 100 g of cream
- The other ingredients are :
Octyldodecanol, light liquid kerosene, stearyl alcohol*, cetyl alcohol*, myristic alcohol, polysorbate 60 (E435)*, sorbitan stearate, benzyl alcohol (E1519)*, lactic acid (E270), purified water.
*See section 2 "MYCOSTER contains".
What MYCOSTER 1 per cent cream is and what it contains
This medicine comes in the form of a homogeneous, virtually white cream in a flexible, white, sealed 30g aluminum tube with a polyethylene screw cap.
Marketing authorization holder
PIERRE FABRE MEDICAMENT
LES CAUQUILLOUS
81500 LAVAUR
Marketing authorization holder
PIERRE FABRE MEDICAMENT
PARC INDUSTRIEL DE LA CHARTREUSE
81100 CASTRES
Manufacturer
PIERRE FABRE MEDICAMENT PRODUCTION
SITE PROGIPHARM
RUE DU LYCEE
45500 GIEN
Product names in member states of the European Economic Area
Not applicable.
The last date on which this leaflet was revised was :
[to be completed subsequently by the holder].
Other
Detailed information on this drug is available on the ANSM (France) website.