NOTICE
ANSM - Updated on : 05/04/2016
Product name
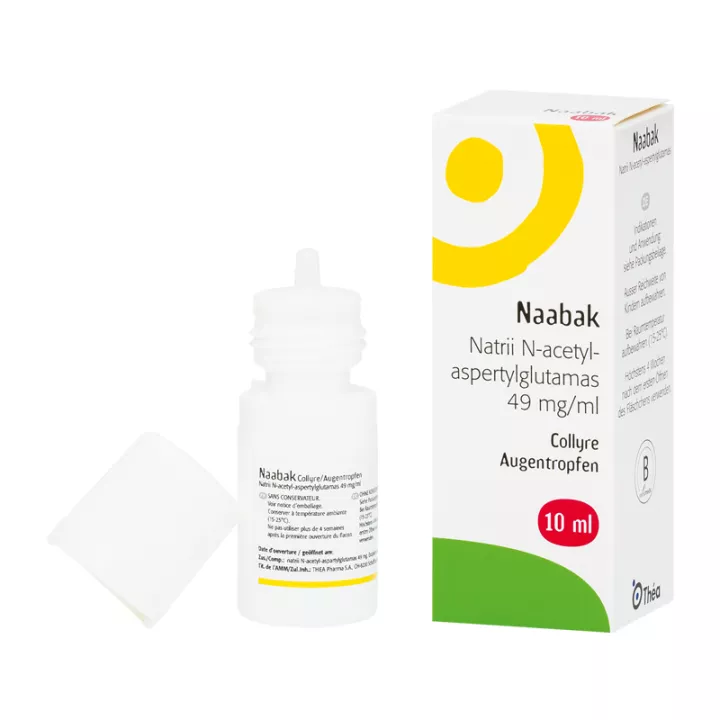
NAABAK 4.9 PER CENT, eye drops solution
N-acetyl aspartyl glutamic acid sodium
Box
Please read this leaflet carefully before using this medicine. It contains important information for your treatment.
If you have any further questions, please ask your doctor or pharmacist.
- Keep this leaflet as you may need to read it again.
- If you need further information or advice, ask your pharmacist.
- If symptoms worsen or persist, consult a doctor.
- If you notice any side effects not mentioned in this leaflet, or if you experience any of the effects mentioned as serious, please tell your doctor or pharmacist.
Contents
In this leaflet :
1. WHAT IS NAABAK 4.9 PER CENT, eye drops solution AND WHAT IS IT USED FOR?
2. WHAT DO YOU NEED TO KNOW BEFORE USING NAABAK 4.9 PER CENT, eye drops solution?
3. HOW DO I USE NAABAK 4.9 PER CENT, eye drops solution?
4. WHAT ARE THE POSSIBLE SIDE EFFECTS?
5. HOW DO I USE NAABAK 4.9 PER CENT, eye drops solution?
6. ADDITIONAL INFORMATION
1. WHAT IS NAABAK 4.9 PER CENT, eye drops solution AND WHAT IS IT USED FOR?
Pharmacotherapeutic class
OCULAR ANTI-ALLERGY
(S = sense organ)
Therapeutic indications
To relieve ocular symptoms of allergic origin (conjunctivitis, blepharoconjunctivitis).
2. WHAT DO I HAVE TO KNOW BEFORE USING NAABAK 4.9 PER CENT, eye drops solution?
List of information required before use
Not applicable.
Contraindications
Never use NAABAK 4.9 PER CENT, eye drops solution:
- if you are allergic (hypersensitive) to N-acetyl aspartyl glutamic acid or to any of the other ingredients of NAABAK 4.9 PER CENT, eye drops.
IF IN DOUBT, ASK YOUR DOCTOR OR PHARMACIST FOR ADVICE.
Precautions for use; special warnings
Take special care with NAABAK 4.9 PER CENT, eye drops solution:
Special warnings
Do not inject, do not swallow
The absence of preservatives in instilled drops means that soft contact lenses can be worn.
Do not touch the eye with the tip of the bottle.
Precautions for use
- For children under 4 years of age, DO NOT USE WITHOUT MEDICAL ADVICE.
- In case of severe conjunctival allergy, CONSULT YOUR DOCTOR.
- Do not exceed the prescribed or recommended dosage, and do not interrupt your treatment without medical advice.
IF IN DOUBT, ASK YOUR DOCTOR OR PHARMACIST FOR ADVICE.
Interactions with other drugs
Use of other drugs
If you are currently using (or plan to use) another eye drop, wait 15 minutes between instillations.
Talk to your doctor or pharmacist if you are taking or have recently taken any other medication, including over-the-counter medicines.
Interactions with food and drink
Not applicable.
Interactions with herbal products or alternative therapies
Not applicable.
Use during pregnancy and breast-feeding
Pregnancy and breast-feeding
IN GENERAL, ALWAYS SEEK THE ADVICE OF YOUR DOCTOR OR PHARMACIST BEFORE TAKING ANY MEDICATION DURING PREGNANCY OR WHILE BREAST-FEEDING.
Sportsmen
Not applicable.
Effects on ability to drive vehicles or use machines
Not applicable.
List of excipients
Not applicable.
3. HOW TO USE NAABAK 4.9 PER CENT, eye drops solution
Instructions for proper use
Not applicable.
Dosage, Method and/or route(s) of administration, Frequency of administration and Duration of treatment
Dosage
1 drop, 2 to 6 times a day.
Method and route of administration
Ophthalmic route.
To ensure proper use of eye drops, certain precautions must be taken:
- Wash hands thoroughly before use,
- Do not touch the eye or eyelids with the tip of the bottle.
Instill one drop of eye drops into the conjunctival cul de sac, pulling the lower eyelid slightly upwards.
The time it takes for a drop to appear is longer than with conventional eye drops.
Recap bottle after use.
Frequency of use
On average, 2 to 6 times a day.
Treatment duration
1 vial of 5 ml provides approximately 20 days' treatment at the recommended average dosage.
Once symptoms have improved, treatment should be continued for the duration of the allergy risk period.
Symptoms and instructions in case of overdose
If you have taken more NAABAK 4.9 PER CENT, eye drops solution than you should have :
In case of overdose, rinse with sterile saline solution.
Instructions in case of missed dose(s)
Not applicable.
Risk of withdrawal syndrome
Not applicable.
4. WHAT ARE THE POSSIBLE SIDE EFFECTS?
Description of undesirable effects
As with all medicines, this drug may cause undesirable effects, but they do not occur systematically in everyone.
- Possibility of brief burning or tingling sensations on instillation.
Reporting side effects
This also applies to any side effects not mentioned in this leaflet. You can also report adverse reactions directly via the national reporting system: Agence nationale de sécurité du médicament et des produits de santé (Ansm) and the network of Centres Régionaux de Pharmacovigilance - Website: www.ansm.sante.fr.
By reporting adverse reactions, you contribute to providing more information on drug safety.
5. HOW TO USE NAABAK 4.9 PER CENT, eye drops solution?
Keep out of the reach and sight of children.
Expiration date
Do not use NAABAK 4.9 PER CENT, eye drops after the expiration date stated on the outer packaging.
Storage conditions
Store at a temperature not exceeding 25°C.
After first opening: eye drops can be stored for 3 months.
If necessary, warnings against certain visible signs of deterioration
Medicines must not be disposed of in the sewage system or with household waste. Ask your pharmacist what to do with unused medicines. These measures will help protect the environment.
6. FURTHER INFORMATION
Complete list of active ingredients and excipients
What does NAABAK 4.9 PER CENT, eye drops solution contain?
The active ingredients are
N-acetyl aspartyl glutamic acid sodium ........................................................................................... 4.9 g
per 100 ml solution.
Other ingredients are:
Excipients: sodium hydroxide or hydrochloric acid, water for injection.
Pharmaceutical form and contents
What is NAABAK 4.9 PER CENT, eye drop solution and what does it contain?
This medicine is presented as an eye drop in a 5 or 10 ml bottle.
Name and address of the marketing authorization holder and of the manufacturing authorization holder responsible for batch release, if different
Holder
Laboratoires THEA
12, rue Louis Blériot
63017 Clermont-Ferrand Cedex 2, France
Operator
Laboratoires THEA
12 Rue Louis Blériot
ZI du Brezet
63100 CLERMONT FERRAND
Manufacturer
LABORATOIRES THISSEN
Rue de la Papyrée 2-4
1420 BRAINE-L'ALLEUD
BELGIUM
or
EXCELVISION
Rue de la Lombardière
07100 ANNONAY
or
FARMILA-THEA FARMACEUTICI SPA
Via E. Fermi 50
20019 SETTIMO MILANESE (MI)
ITALY
Product names in member states of the European Economic Area
Not applicable.
Date of approval of the package leaflet
This leaflet was last approved on {date}.
MA under exceptional circumstances
Not applicable.
Internet information
Detailed information on this drug is available on the INSM (France) website.
Information reserved for healthcare professionals
Not applicable.
Other
Not applicable.