NOTICE
ANSM - Updated on : 30/12/2024
Product name
MYCOSTER 8%, film-forming solution for cutaneous application in bottle
Ciclopirox
Box
Please read this leaflet carefully before using this medicine, as it contains important information for you.
You must always use this medicine in strict accordance with the information provided in this leaflet or by your doctor or pharmacist.
- Keep this leaflet. You may need to read it again.
- Ask your pharmacist for any advice or information you may need.
- If you experience any side effects, talk to your doctor or pharmacist. This also applies to any side effects not mentioned in this leaflet. See section 4.
- You should contact your doctor if you do not feel any improvement or if you feel less well.
What does this leaflet contain?
1. What is MYCOSTER 8%, film-forming solution for cutaneous application in a bottle, and what is it used for?
2. What do I need to know before using MYCOSTER 8%, film-forming solution for cutaneous application in a bottle?
3. How do I use MYCOSTER 8%, film-forming solution for cutaneous application in a bottle?
4. What are the possible side effects?
5. How do I keep MYCOSTER 8%, film-forming solution for cutaneous application in a bottle?
6. Package contents and other information.
1. WHAT IS MYCOSTER 8%, film-forming solution for cutaneous application in bottles AND WHAT IS IT USED FOR?
Pharmacotherapeutic class: Other topical antifungals - ATC code: D01AE14.
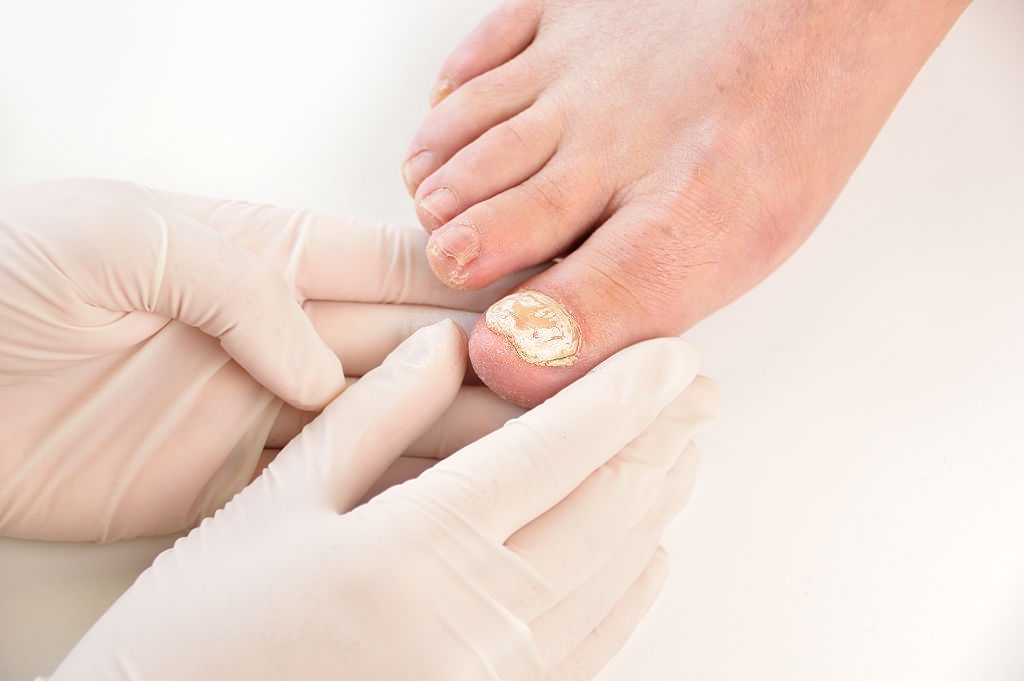
MYCOSTER is indicated in adults for the treatment of fungal infections (onychomycosis) of the toenails and fingernails. The active substance, ciclopirox, penetrates the nail and destroys the fungi (antifungal).
You should contact your doctor if you do not feel any improvement or if you feel less well.
2. WHAT DO I HAVE TO KNOW BEFORE USING MYCOSTER 8%, film-forming solution for cutaneous application in a bottle?
Never use MYCOSTER 8%, film-forming solution for cutaneous application in bottles:
- if you are allergic to ciclopirox or any of the other ingredients listed in section 6.
Warnings and precautions
Talk to your doctor or pharmacist before using MYCOSTER.
Be careful with MYCOSTER :
- Do not swallow
- Avoid contact with eyes and mucous membranes until MYCOSTER has dried.
- MYCOSTER should only be used topically.
- Do not apply ordinary nail varnish or other nail cosmetics to treated nails.
- In the event of an allergic reaction, stop treatment immediately, carefully remove the nail varnish with a nail polish remover and contact your doctor.
- If you have several nails to treat (more than 5) or if you have diabetes or an immune disease, contact your doctor, as you may need additional oral treatment.
- If your nail has become detached during cleaning, particularly if you have diabetes or diabetic neuropathy (nerve damage), consult your doctor.
Children
The safety and efficacy of MYCOSTER in neonates, infants and children have not been established.
Other drugs and MYCOSTER 8%, film-forming solution for cutaneous application in bottle
Tell your doctor or pharmacist if you are taking, have recently taken or may take any other medicines.
MYCOSTER 8%, film-forming solution for cutaneous application in bottles with food and drink
Not applicable.
Pregnancy and breast-feeding
Pregnancy
If you are pregnant or breast-feeding, think you may be pregnant or are planning to become pregnant, ask your doctor or pharmacist for advice before taking this medicine.
This medicine may be used by pregnant women.
Breast-feeding
There are no adequate data on the excretion of ciclopirox into breast milk. A risk to the newborn/infant cannot be excluded. Consequently, MYCOSTER should not be used during lactation without medical advice.
Driving and use of machinery
MYCOSTER has no effect on the ability to drive and use machines.
MYCOSTER 8%, film-forming solution for cutaneous application in bottle contains
Not applicable.
3. HOW DO I USE MYCOSTER 8%, film-forming solution for cutaneous application in a bottle?
Always use this medicine exactly as instructed in this leaflet or as directed by your doctor or pharmacist. Check with your doctor or pharmacist if in doubt.
Route of administration
Dermal route.
For external use only.
For adult use only.
Dosage
The recommended dose is one application per day, preferably in the evening.
Close the bottle tightly after each use.
After each use, observe storage conditions (see paragraph 5).
Before starting MYCOSTERtreatment: remove as much diseased tissue as possible from the nail with scissors, nail clippers or a nail file.
For theduration of treatment:
- Apply a thin layer of MYCOSTER to the entire surface of all diseased nails once a day, preferably in the evening.
- Once a week, before applying MYCOSTER, remove the nail polish layer with a commercial nail polish remover.
- Each time, remove as many diseased nail tips as possible.
Duration of treatment
Treatment should continue until complete healing and regrowth of healthy nail tips.
Depending on the severity of your infection (extensive involvement of one or more fingernails and toenails), contact your doctor as you may need further oral treatment.
Treatment usually lasts between 3 months (fingernail fungus) and 6 months (toenail fungus). Treatment should not exceed 6 months.
If in doubt about continuing treatment, ask your doctor or pharmacist for advice.
If you have used more MYCOSTER 8%, film-forming solution for cutaneous application in bottles than you should have, contact your doctor or pharmacist.
Not applicable.
If you forget to use MYCOSTER 8%, film-forming solution for cutaneous application in bottles
Do not use a double dose to make up for the one you forgot to apply.
Continue treatment in accordance with the instructions for use given earlier in this section.
If you stop using MYCOSTER 8%, film-forming solution for cutaneous application in bottle
If you stop or interrupt treatment too soon, your mycosis may reappear. If you're not sure whether your skin condition has improved, ask your doctor or pharmacist for advice.
If you have any further questions about the use of this medicine, please ask your doctor or pharmacist.
4. WHAT ARE THE POSSIBLE SIDE EFFECTS?
Like all medicines, this drug may cause undesirable effects, but they do not occur systematically in everyone.
Undetermined frequency (cannot be estimated on the basis of available data)
- Contact dermatitis (eczema-like rash).
- Generalized allergic reactions (hypersensitivity), which may be severe, in which you may experience swelling (edema), itching (pruritus), an itchy or non-itchy rash (urticaria) or difficulty breathing (dyspnea). If you experience any of these effects, you should stop treatment immediately and contact a doctor as soon as possible.
Reporting side effects
If you experience any side effects, please tell your doctor or pharmacist. This also applies to any side effects not mentioned in this leaflet. You can also report adverse reactions directly via the national reporting system: Agence nationale de sécurité du médicament et des produits de santé (ANSM) and the network of Centres Régionaux de Pharmacovigilance - Website: https://signalement.social-sante.gouv.fr/.
By reporting adverse reactions, you are helping to provide more information on drug safety.
5. HOW TO CONSERVE MYCOSTER 8%, film-forming solution for cutaneous application in a bottle?
Keep out of sight and reach of children.
Do not use this medicine after the expiration date indicated on the bottle after EXP. The expiration date refers to the last day of that month.
Store in original, light-resistant packaging.
After first opening, keep the bottle tightly closed in the outer packaging.
After first opening, store for a maximum of 3 months.
Do not dispose of any medicines in the sewage or household waste. Ask your pharmacist to dispose of any medicines you no longer use. This will help protect the environment.
6. CONTENTS AND OTHER INFORMATION
What MYCOSTER 8%, film-forming solution for cutaneous application in bottle, contains
- The active substance is :
Ciclopirox................................................................................................................................... 8 g
Per 100g of film-forming solution
- The other components are :
Copolymer of methyl vinyl ether and maleic acid monobutyl ester, ethyl acetate, isopropanol.
What MYCOSTER 8%, film-forming solution for cutaneous application in a bottle, is and what it contains
This medicine takes the form of a clear, slightly viscous, colorless to slightly brownish-yellow liquid.
Box of 1 bottle with brush.
Marketing authorization holder
PIERRE FABRE MEDICAMENT
LES CAUQUILLOUS
81500 LAVAUR
Marketing authorization holder
PIERRE FABRE MEDICAMENT
PARC INDUSTRIEL DE LA CHARTREUSE
81100 CASTRES
Manufacturer
PIERRE FABRE MEDICAMENT PRODUCTION CAHORS
SITE CAHORS
LE PAYRAT
46000 CAHORS
Product names in member states of the European Economic Area
Not applicable.
The last date on which this leaflet was revised was :
[to be completed subsequently by the holder].
Other
Detailed information on this drug is available on the ANSM (France) website.
ADVICE AND RECOMMENDATIONS TO ACCOMPANY YOUR TREATMENT
Lonychomycosis: what are the risk factors?
For toenails:
- Repeated microtrauma to the foot or toes, toe malposition.
- Wearing closed and/or plastic shoes or synthetic socks, which promote maceration.
- Exposed occupations requiring the wearing of boots or protective footwear that facilitate perspiration.
- Barefoot sports: swimming, judo, marathon running.
For fingernails:
Look for foot fungus (dermatophytosis).
Repeated contact with water (candidiasis).
Trauma or irritation of the nail by :
- Repeated microtrauma (gardening),
- Manual use of detergents (laundry and other corrosive products),
- Exposed professions (hairdressers, manicurists, chiropodists).
Lonychomycosis: how to prevent and avoid contamination?
- Disinfect socks and shoes. Ask your pharmacist for advice.
- Treat any damage to the skin between the toes or on the foot as quickly as possible.
- Use individual towels toavoid contaminating other people.
- Thoroughlydry the spaces between toes and the whole foot after showering or bathing to avoid contamination of other nails and skin.
Lonychomycosis: why is it important to follow treatment?
To be effective, treatment must be taken for the full duration of the treatment, without interruption, until the nail has completely regrown.
DON'T BE DISCOURAGED!
It can take 3 months for fingernails and 6 months for toenails.
All treatments are long because it's the time it takes for healthy nails to grow back that shows their effectiveness.